Written by:
Andrea Jordà, Project Engineer at Klinea Biotech & Pharma Engineering
Jordi Gibert, Head of the Biotechnology Unit at Klinea Biotech & Pharma Engineering
Biotechnology has emerged as a key driver in the transformation of the pharmaceutical industry, revolutionising the way medicines are discovered, developed and produced. In a context where complex diseases and global health challenges are becoming increasingly challenging, biotechnology offers innovative and effective solutions.
One of the most notable advances that biotechnology has brought to the pharmaceutical industry is the development of biological drugs. While traditional medicines, generally based on chemical compounds, offer more generalised treatments, biological drugs are products developed from living organisms, which allows them to interact with the body in a more precise manner. This results in therapies that are not only more specific and effective, but also in solutions that tend to have fewer side effects.
Furthermore, biotechnology’s recent ability to design personalised treatments has revolutionised the approach to medicine, allowing therapies to be tailored to individual patient characteristics. This approach is particularly crucial in the treatment of chronic, genetic and oncological diseases, where responses to treatment can vary between patients.
In 2023, the global biopharmaceutical market reached revenues of approximately £373.60 billion, and is expected to grow to over £856.10 billion by 2030 [1]. Within this market, the monoclonal antibody sector leads with 42% of the total, driven by its growing use in cancer treatment due to its ability to attack cancer cells with complete specificity.
Large biopharmaceutical companies are not only transforming the world of healthcare with their innovative technologies, but are also driving a multi-billion pound market. As demand for biologics grows exponentially, the question arises: which companies are at the forefront of this revolution?
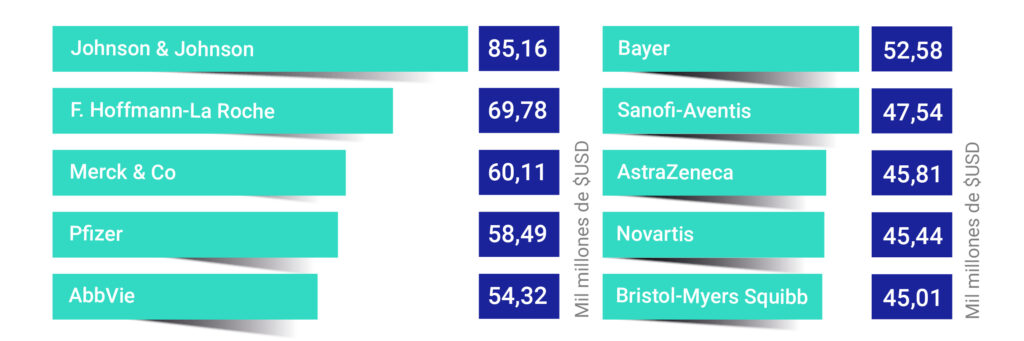
Figure 1. Annual revenue in 2023 of the leading biopharmaceutical companies [2]
The global market for biological drugs has experienced unprecedented growth. More than 50% of the top-selling drugs in 2023 were obtained using biotechnological technologies. Messenger RNA (mRNA)-based COVID-19 vaccines, such as those developed by Pfizer/BioNTech and Moderna, have been key to this growth and the adoption of new biotechnological technologies.
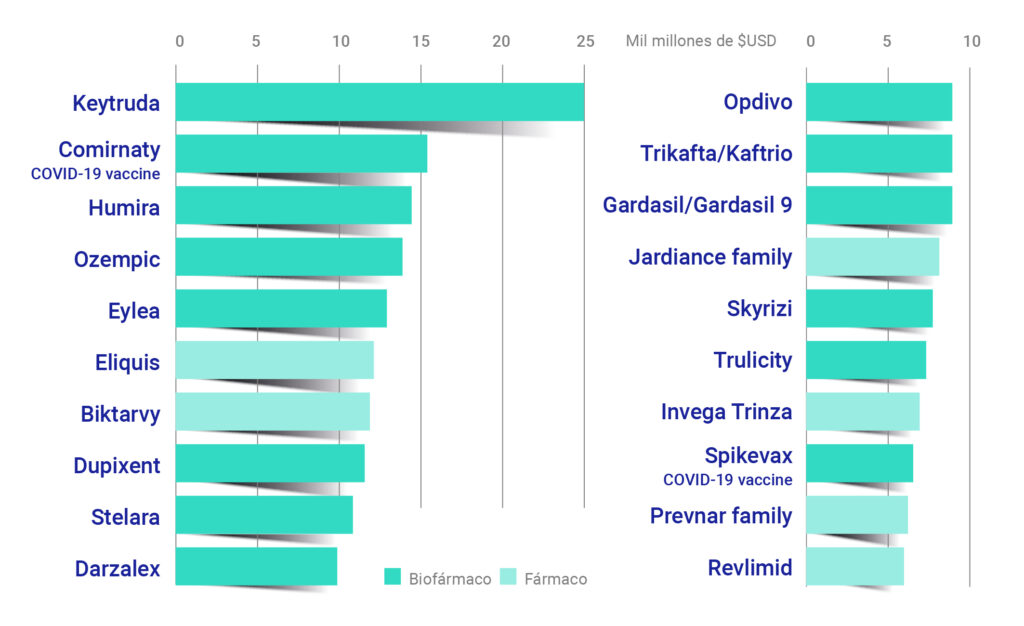
Figure 2. The 20 pharmaceutical products with the highest sales volume in 2023. [3]
Although biological drugs currently dominate the pharmaceutical industry, their leadership could be affected in the coming years due to the expiry of numerous patents and the arrival of biosimilars. These are biological medicines that are equivalent in molecular structure, pharmacological activity, efficacy and safety to another biological medicine known as the reference medicine [4] at a lower cost.
The expiry of patents on best-selling drugs opens the door to these second-generation products, which could drastically alter the dynamics of the market, currently dominated by high prices.
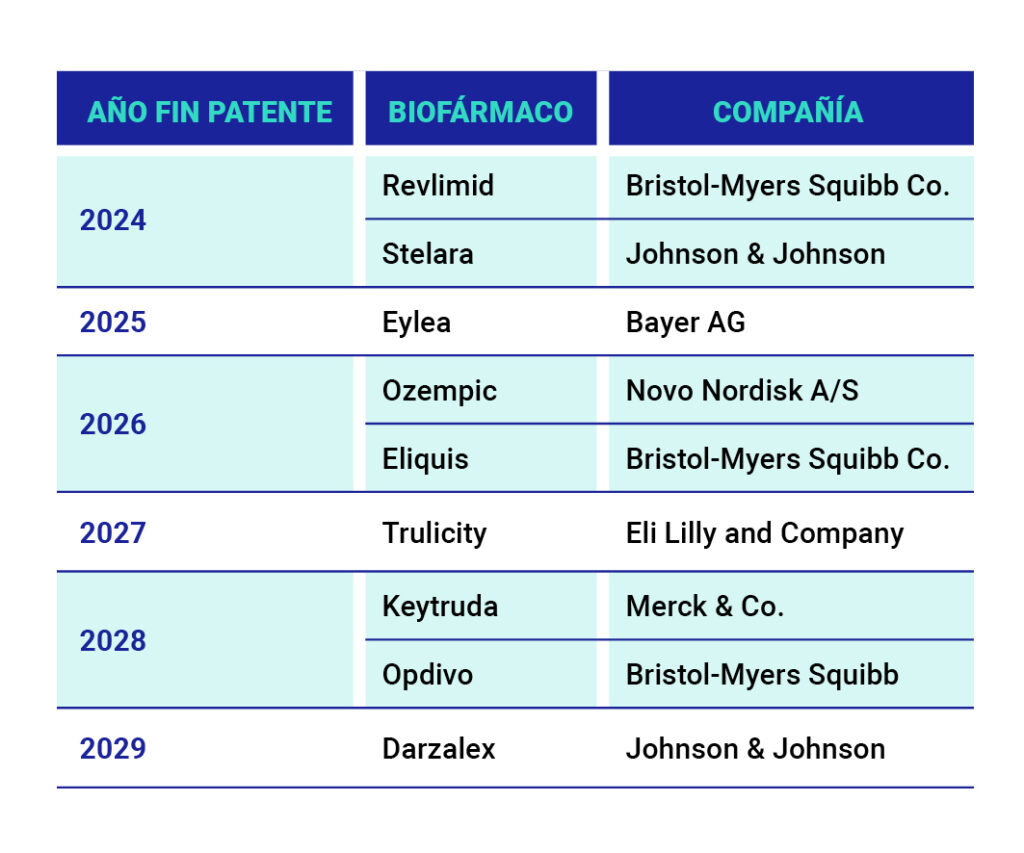
Table 1. Expiry dates of patents for the best-selling drugs. [5]
Investment and pipeline in biotechnology
One of the key factors in the growth of biotechnology within the pharmaceutical industry is the increasing investment in research and development (R&D). It is not surprising that major pharmaceutical companies around the world are increasingly redirecting their R&D focus towards biotechnology-based products, as this not only strengthens their product pipeline, but also allows them to remain competitive in an ever-evolving market.
In 2022, the top 15 pharmaceutical companies spent more than $138 billion on R&D, an increase of 44% compared to previous years. This remarkable expenditure marks the industry’s commitment to innovation and the advancement of new therapies [6].
The success of a biopharmaceutical company depends largely on the strength of its pipeline of new products. The greater the investment in the early stages of basic research and preclinical development, the greater the likelihood of identifying promising compounds that can advance to clinical trials.
In 2024, biological drugs had a record year, with nine new biologics approved in the first quarter, surpassing the five approvals in the same period in 2023 [7].
Furthermore, as part of their strategy, large biopharmaceutical companies acquire small companies and start-ups that have developed drugs with the potential to become blockbusters (products with more than £1 billion in annual revenue). This allows them to quickly access advanced innovations without fully assuming the risk of initial development.
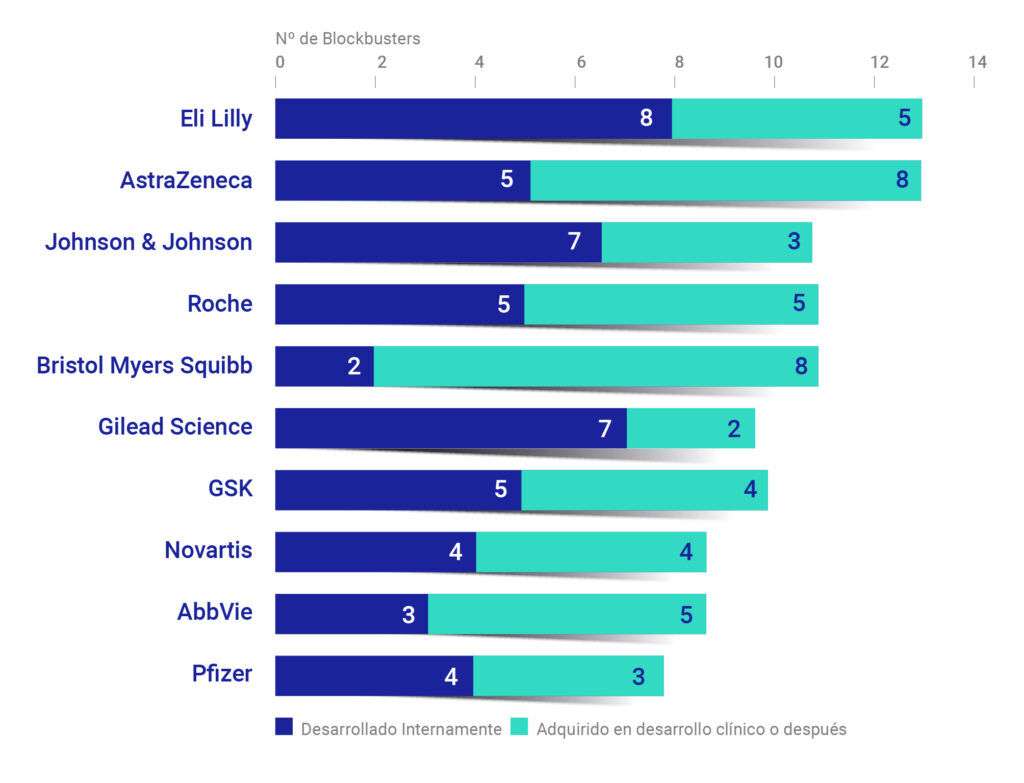
Figure 3. Companies with the most blockbusters approved, 2014–2023.[8]
Advanced therapies: the future of medicine
Advanced therapies have represented a disruptive change in modern medicine, offering long-term treatments or even definitive cures for diseases that until recently had no treatment. These therapies mainly include three key areas: cell therapy, gene therapy, and tissue engineering products.
The advanced therapies market has shown significant growth in recent years, reaching global revenues of approximately £11.99 billion in 2023, and is projected to exceed £35.59 billion by 2032.[9].
Regulatory approvals have helped consolidate growth in this sector. Over the last decade, several advanced therapy products have been approved worldwide, such as Kymriah and Yescarta, CAR-T treatments targeting certain types of cancer, and Luxturna, a gene therapy approved for hereditary retinal dystrophy. These approvals have generated considerable interest among pharmaceutical companies and opened up a new line of business for them.
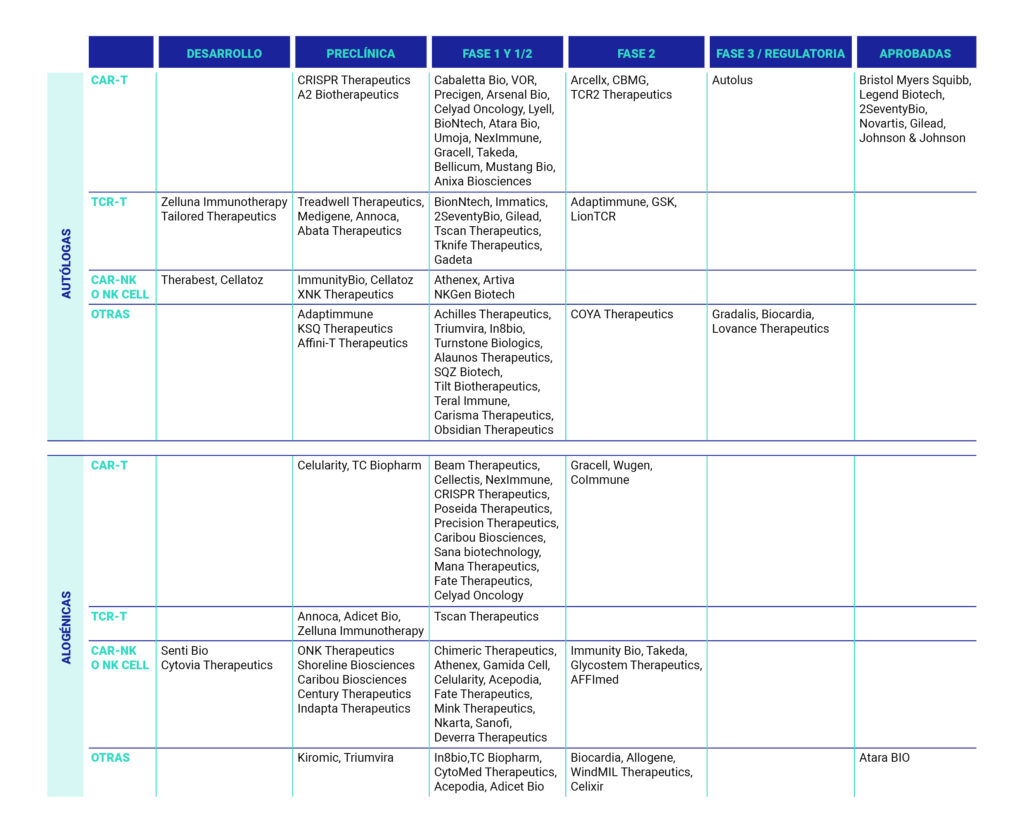
Table 2. Innovations in cell therapies. [10]
However, despite the great opportunities offered by these therapies, one of the main challenges they face is their high cost. The price of a treatment based on advanced therapies can exceed several million dollars per patient, raising questions about their long-term accessibility and availability. This makes it essential to change the approach to the production of these therapies and to promote the development of automated systems that guarantee the accessibility of these drugs for the whole of society.
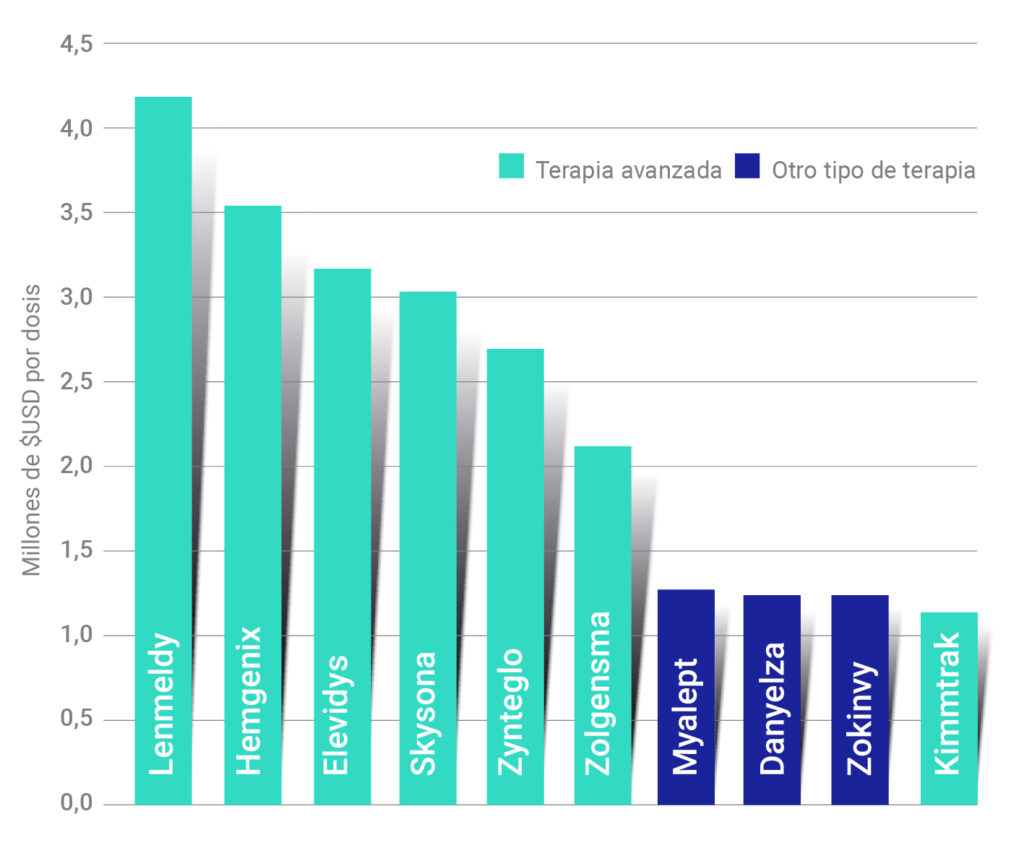
Figure 4. The 10 most expensive biological therapies per dose. [11]
The advanced therapies market will mark a turning point for the biopharmaceutical industry in the coming years. By ensuring continuous advancement in production equipment technologies and promoting the development of fully automated equipment to improve production efficiency and consistency, it is possible that these therapies will become more accessible, prices per dose will be reduced, and a greater number of patients will benefit globally.
Conclusions
The future of biotechnology in the pharmaceutical industry is promising. With continued advancement and investment in biotechnology, the number of diseases treatable with biological therapies will increase. Even definitive solutions for chronic and genetic diseases will be offered through cell and gene therapy.
Although challenges remain in terms of cost and accessibility, biological drugs represent one of the most promising areas in modern medicine.
At Klinea, as specialists in biopharmacy and biological processes, we want to help our clients maximise the impact of their products. Contact us to learn more about biosimilars, biological processes and biotechnology: klinea@klinea.eu
Bibliography
[1] Research, P. (2023, 4 agosto). Biopharmaceuticals Market Revenue to Reach USD 8,56,100 Million by 2030. Yahoo Finance. https://finance.yahoo.com/news/biopharmaceuticals-market-revenue-reach-usd-150000985.html?guce_referrer=aHR0cHM6Ly93d3cuYmluZy5jb20v&guce_referrer_sig=AQAAAAJd3_qNzVuOTvlOeTXFP92f8Bo7ZxHDvyfy36nVX9ubD_Pv_CAFzrWCBIMVaA6GqrRRiVh4Ve-7dFYEMpF36PX971FcgNqzPqU_vuAriSA_RpnqQajwjz30jaaIva3DZ-Fhw2WJMgo-A_6xu0VDBQRJvO43DZRs8NnDxzT_h0O3&guccounter=2
[2] Editor. (s. f.). Top 20 Biopharma Companies of 2024. Pharmashots. https://pharmashots.com/17231/top-20-biopharma-companies-of-2024
[3] Buntz, B. (2024, 21 mayo). Best-selling pharmaceuticals of 2023 reveal a shift in pharma landscape. Drug Discovery And Development. https://www.drugdiscoverytrends.com/best-selling-pharmaceuticals-2023/
[4] Biosimilares. (2024, 16 octubre). Agencia Española de Medicamentos y Productos Sanitarios. https://www.aemps.gob.es/medicamentos-de-uso-humano/medicamentos-biologicos/biosimilares/
[5] GreyB, T. (2025, 27 febrero). List – 31 Biologics Coming Off Patents Between 2022 to 2027. GreyB. https://www.greyb.com/blog/biologics-patents-expiring/
[6] Global Trends in R&D 2022. (s. f.). IQVIA. https://www.iqvia.com/insights/the-iqvia-institute/reports-and-publications/reports/global-trends-in-r-and-d-2022
[7] Research, C. F. B. E. A. (2025, 7 febrero). 2024 Biological License application approvals. U.S. Food And Drug Administration. https://www.fda.gov/vaccines-blood-biologics/development-approval-process-cber/2024-biological-license-application-approvals
[8] Evaluate pharma
[9] Research, S. (s. f.). Advanced Therapy Medicinal Products (ATMP) Market Size by 2032. https://straitsresearch.com/report/advanced-therapy-medicinal-products-market
[10] Anderson, L. A. (2024, 1 abril). 10 of the Most Expensive Drugs in the U.S. Drugs.com. https://www.drugs.com/article/top-10-most-expensive-drugs.html
[11] Anderson, L. A. (2024, 1 abril). 10 of the Most Expensive Drugs in the U.S. Drugs.com. https://www.drugs.com/article/top-10-most-expensive-drugs.html

