Written by:
Jordi Gibert, head of the Biotechnology Unit at Klinea Biotech and Pharma engineering
Albert Canet, project engineer at Klinea Biotech and Pharma engineering
Francisco Valero, coordinator of the bioprocess engineering and applied biocatalysis group.
Genetically modifying a cell to produce a biotechnological product foreign to its own nature, producing it more efficiently compared to native organisms that produce it naturally, or for the cell to produce a biotechnological product modified with respect to its original structure: this is a common tool used for the production of the vast majority of biopharmaceuticals, if not “almost” all, thanks to recombinant DNA technology, developed in the 70s of the last century.
As a result, monoclonal antibodies, state-of-the-art vaccines, blood factors, growth factors, hormones and other recombinant enzymes and proteins for therapeutic use are now commercially available.
The use of these biopharmaceuticals has become widespread, reaching an estimated market value of US$617 billion in 2024, already representing a significant share of the US$1747 billion total market value of pharmaceuticals in 2024, and with a prospect of their market doubling by 2032 [1,2].

Tabla 1. Examples of top biopharmaceutical products by sales volume in 2015; for more information on biopharmaceutical products see references [6,7,8].
One of the first of these biopharmaceuticals in commercial use was insulin produced by escherichia coli bacteria, approved by the FDA in 1982 [3]. This first step demonstrated that the use of microorganisms – such as bacteria, mainly E. coli, and yeasts, such as Sacchareomyces cerevisiae and Pichia pastoris – could be extended to the production of a wide range of biopharmaceuticals [4].
In general, microbial platforms have higher growth rates than mammalian cells and can reach higher cell densities [4], reaching higher productivities; these advantages make microbial platforms cost-efficient cell factories compared to mammalian cell platforms in the production of biopharmaceuticals – and in general in the production of recombinant compounds for use in other sectors beyond pharmaceuticals -. However, the lack or poor ability of bacteria and yeasts to perform post-translational modifications of bioproducts identical to that of higher organisms has limited their use in the production of several biopharmaceuticals, with mammalian cells being the most widely used for this type of bioproducts; these modifications – also known as “humanizations”, such as the correct glycosylation sequence – are necessary for the correct functioning of some molecules and/or their rejection by the immune system.
The trend in recent years is that mammalian cell platforms have gained ground over microbial ones in the production of biopharmaceuticals – especially due to the irruption and rise of monoclonal antibodies, which require post-translational modifications [5]. This is demonstrated by the numbers of biopharmaceutical approvals: until 1989 mammalian cell platforms accounted for 33% of the total, whereas in the period 2015-2018 they accounted for 79% [5]. In market volume already in 2015 only 4 of the top 10 biopharmaceuticals were produced by microbial platforms, as shown in Table 1 [3]. However, where the use of microorganisms still has a prominent role is in the production of proteins, blood factors, hormones, growth factors, interferons, interleukins, tumor factors and vaccines [5]. In addition, it should be mentioned that the use of plasmids for different therapies – such as gene therapies – also drives the use of microbial platforms, as they are produced with microorganisms.
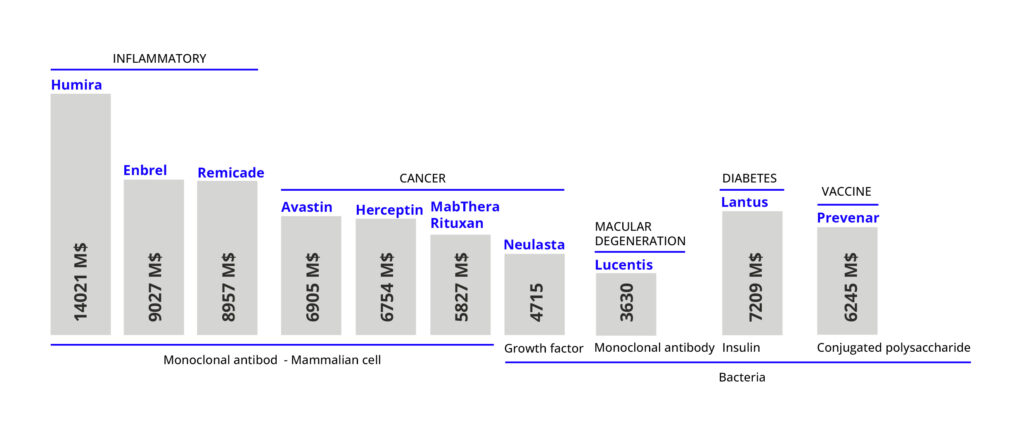
Figure 1. Laboratory analysis of biological samples
Currently there are several research groups developing cellular factories based on microbial platforms – through genetic modifications – with the ability and/or improvement in post-translational modifications that can incorporate the glycosylation pattern characteristic of the mammalian cellsas well as improvements in other no less important problems such as secretion and folding of the synthesized molecules [5,6]; for example, E. coli is commonly known for having the poor folding of the synthesized moleculesThe glycosylation of P. pastoris yeast, which is known as inclusion bodies, is a process that requires additional processing of these insoluble bodies to obtain the final recombinant product in an active form. In the case of P. pastoris yeast, there are modified commercial strains with the same glycosylation pattern as humans, such as the Pichia GlycoSwith® platform.
On the industrial operational and engineering side, the two platforms – i.e. mammalian cells and microorganisms – present the same bioprocess structure: first cell growth, associated to the production of the molecule – known as upstream -, and the subsequent recovery and purification of the molecule – known as the downstream -. However, microbial platforms have shorter production times and high cell density systems, making the processes more economical compared to mammalian cells, as discussed above – in addition to this, the culture media for mammalian cells are usually much more expensive than those for bacteria and yeast -.
In terms of operations, certainly in the downstream downstream is where there are more differences between the two platforms: in productions with microorganisms, in some cases there are protein solubilization and folding stages, as is the case with E. coli discussed above, whereas in mammalian cells, viral inactivation steps are often required. However, in general, production with CHO – the main mammalian cell platform for biopharmaceutical production – presents a downstream simpler and/or more efficient than microbial platforms, since the synthesis and expression of biomolecules is purer [5].
On the other hand, if the engineering characteristics of the equipment used are analyzed, in general, their attributes are similar – i.e. i.e. agitation, oxygen supply, temperature control, among others, although with different ranges of values. A good example of this is the irruption of the technologies single useespecially in bioreactors; single-use bioreactors single use bioreactors are of great utility for mammalian cell platforms – in fact, the vast majority of bioreactors used in mammalian cells today are single use -However, they tend to have a low matter transfer – they admit little agitation – and a low heat transfer – they are materials with poor thermal conduction – which limit their use in microbial platforms. However, bioreactors are already available. single use improved by applying them to the production of bacteria and yeast in cell factories.
Although microbial platforms have biological limitations and are unable to produce certain biopharmaceuticals, at a technical level their productivity exceeds that of mammalian cell platforms. Scientific research in the production of biopharmaceuticals with microorganisms aims to overcome these biological limitations.
If you are interested in learning more about biopharmaceutical production facilities and how we can help you, please contact us: klinea@kliena.eu.
Bibliography
1. Biopharmaceuticals Market Size, Share & Industry, By Type, By Application, By Distribution Channel and Regional Forecast 2025-2032; Fortune Business Insights; 2025
2. Pharmaceutical Drugs Global Market Report 2025; The Business Research Company; 2026
3. Biopharmaceuticals from microorganism: from production to purification: Angela Faustino Jozala et al.; Brazilian Journal of Microbiology; 2016.
4. Whitepaper: Advancements in microbial manufacturing of biopharmaceuticals; Boehringer Ingelheim; 2024
5. Microbial protein cell factories fight back ?; Lukas A. Rettenbacher; Trends in Biotechnology; 2022
6. Production of Biopharmaceuticals in E. coli: Current Scenario and Future Perspectives; Mohammed N. Baeshen et al.; Journal and Microbiology and Biotechnology; 2015.
7. Yeast as Biopharmaceutical Production Platforms; Natalja Kulagina et al.; Frontiers in Fungal Biology; 2021.
8. Evolving Paradigms of Recombinant Protein Production in Pharmaceutical Industry: A Rigorous Review; Achuth Jayakrishnan et alSci; 2024