For some years now, we have seen “stem cells” more and more in the news. They are supposed to be capable of extraordinary things, to be a real revolution and to have the potential to improve the lives of many people. Yes, all of the above is true. In fact, it is already a reality today. But the potential of this technology is immense. And it can take us much, much further than it already has.
What are stem cells?
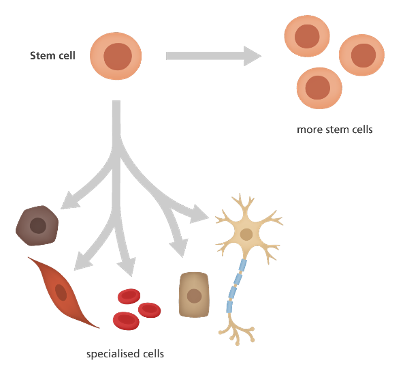
But let’s start with the basics. What are stem cells? ? Imagine you could get only from one material everything you need to build a house: wood, cement, iron, stone, etc. Impressive, isn’t it? Well, that is what stem cells are for the human body.
Stem cells are the precursors of all cell types in our body. They can give rise to more stem cells or differentiate into any type of cell in the body (heart, muscle, blood, bone, etc.).
Yes, you are right: their potential is enormous. That is why they have attracted the attention of the pharmaceutical industry, research groups and regulatory authorities. In fact, the FDA has already approved more than a dozen treatments using stem cells. Considering that ten years ago this list did not exist, we can speculate that it will grow exponentially in the upcoming years.
Why do we talk about stem cells?
Your mind is probably already racing with ideas about what can be done with stem cells. Let’s look at some of these possible uses:
- Therapy to regenerate cellular tissues. Take, for example, a disease caused by the malfunction of a particular type of cell, such as type 1 diabetes. People suffering from this disease are unable to produce insulin due to the destruction of the cells in the pancreas that are responsible for it. If we manage to replace these cells with others that are not destroyed by stem cells, we could put an end to a chronic disease, which nowadays requires continuous treatment for life.
Tissue regeneration using stem cells represents possibly the greatest potential of stem cells. In fact, it is already a reality. It is currently used to treat severe skin lesions or malfunctioning heart cells.
- Reducing donor dependency. Certain medical processes require a stem cell donor (e.g. bone marrow transplantation). The availability of stem cells at any time and without donor dependency could help many, many people who are dependent on donation. However, this would require solving the compatibility problems that arise from using cells that do not come from the patient’s own body.
- Creating synthetic organs. Yes, this is thinking “big”, as organs are extremely complex. But if we know that all the organs of the body can be created from a stem cell in the embryonic stage, why shouldn’t we be able to do the same outside the body? This is precisely the only limitation to creating synthetic organs from stem cells nowadays. It is not known which molecular processes would cause different cells to organise themselves into an organ in an environment outside the foetus.
- To be able to make any cell in the body available in a laboratory without the need for a human extraction. Studying and analysing the interactions of new drugs with cells in the body in an in vitro environment is mandatory during drug development. Stem cells can help obtaining any cell types available without the need to remove them from a person.
All these possible uses of stem cells have a common denominator: a large number of cells are needed. A single dose of stem cells to treat cardiac pathologies requires millions of stem cells. It is not an option to rely on donors to make these quantities available. We need to be able to grow stem cells on an industrial scale.
Why do we want to be able to produce them in a GMP environment?
We have already shown why we need to be able to produce stem cells on a large scale. But quantity is not enough. The uses of these cells require and demand quality. As with any medicine, production must be cGMP-compliant. Producing stem cells in a GMP environment will ensure that they are safe for patients and of a consistent level of quality.
Challenges of GMP stem cell production
- Reducing operator manipulation. As with all new technologies, the production process is still immature. Cooperation between pharmaceutical laboratories, equipment manufacturers and process experts is key to developing new equipment that allows production with minimal operator intervention. Human contact with the product during the production phase is one of the main risks of contamination.
- Robotising the process. In line with the previous point, robots are needed to replace the hands of operators for the manipulation of stem cells. In this way, the process can also be carried out in a closed system.
- Performing the process in a closed system. A closed environment is much safer from a contamination point of view. In addition, it allows the rooms where the equipment is located to have a lower classification. This in turn allows for a huge reduction in production costs related to room ventilation.
The present and future of stem cells is uncertain, but bright and exciting at the same time. We are convinced that their applications will open doors to improve the lives of many people in ways we cannot even imagine today. But science moves fast and the challenges of the future are already a reality today. Stem cells are already being used to treat diseases, and their use in the coming years can only increase.
At Klinea, as an engineering company specialising in the biopharmaceutical sector, we are equally amazed and excited by the world of possibilities that is opening up. Our knowledge of cGMP as well as bioprocesses for the production of biopharmaceuticals makes us an ideal partner to bring the new generation of stem cell-based drugs to the market.

